I participated in the inaugural DIY Science day at the Clifton Institute, led by co-Director Eleanor Harris. It’s an adult-tailored version of the Institute’s Young Naturalists program. Not too many rules: explore the field station, formulate a research question, do some science, and present your results. The vibe is a little like the Serendipity Days practice when I was with NPR’s Digital Media.
As I wasn’t sure how the day would play out, I planned a research question ahead of time: roughly, can we measure any differences between two species of oak (White Oak, Quercus alba, and Northern Red Oak, Q. rubra), as sampled as dead leaves from my home and the Institute? The red oaks are well-known for showing more tannins than the white oaks. Is there a way to measure that difference?
So I brought some leaves picked from my backyard, and then at the field station collected some more in the woods. First complication: I was hoping to also analyze another species from the red oaks, Q. falcata, only to find that there are no records of Southern Red Oak at Clifton.
Second complication: methods and materials for measuring tannins were a bit beyond the capabilities of the research station, or so I surmised. However, some follow-up searching suggests some simpler, alternative methods.
 As a result, staffer Bridget Bradshaw suggested some other tests that might yield some interesting results. She also brilliantly suggested a simple way to make equal measured samples from my dead leaves: 10 punches with a paper hole punch from each leaf. No mussing about with weighing something in the sub-gram range. Samples went into small lidded yogurt jars (apparently well-stocked at the research station, and easy handled by the kiddos).
As a result, staffer Bridget Bradshaw suggested some other tests that might yield some interesting results. She also brilliantly suggested a simple way to make equal measured samples from my dead leaves: 10 punches with a paper hole punch from each leaf. No mussing about with weighing something in the sub-gram range. Samples went into small lidded yogurt jars (apparently well-stocked at the research station, and easy handled by the kiddos).
- Simple chromatography with a bit of acetone and (coffee) filter paper.
- Measuring acidity of samples steeped in water and set in the sun for 30 minutes.
 The chromatography didn’t show any results after the hour or so that we had available. But the pH measurement did return some results, on the face of them counter-intuitive. Here’s our 16 pH specimens, sunning on the Clifton Institute porch.
The chromatography didn’t show any results after the hour or so that we had available. But the pH measurement did return some results, on the face of them counter-intuitive. Here’s our 16 pH specimens, sunning on the Clifton Institute porch.
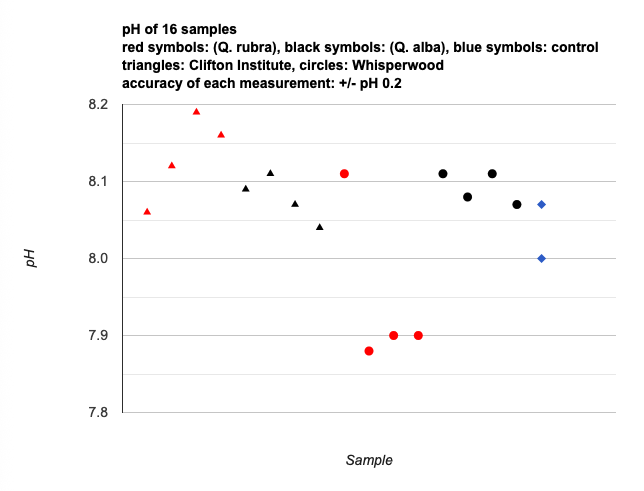
All the White Oaks sampled, as well at the Red Oaks at Clifton, measured more basic than the control (the plain spring water that was used to steep the samples). A possible explanation: fresh oak leaves will measure on the acid end of the scale, but after some decomposition as dead leaves, the acidic compounds leach away preferentially, leaving more basic dead leaf material.
Third complication: the pH meter that we used took several minutes to produce a reading, and readings shifted about as we took our measurements.
For “final presentation” to the group, I drew a quick, crude graph with a Sharpie. I’d like to polish it a bit for presentation here. Update: After some scuffling with the Google Charts API, I managed to produce the following graphic. (It’s a screenshot, not live; the API and vanilla WordPress don’t play well together.)
More research needed: I’d like to try this experiment on some fresh leaves and with a more reliable pH meter.